8-K
0001785279false00017852792026-05-112026-05-11
UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
WASHINGTON, D.C. 20549
FORM 8-K
CURRENT REPORT
Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934
|
Date of Report (Date of earliest event reported): May 11, 2026 |
Metagenomi Therapeutics, Inc.
(Exact name of Registrant as Specified in Its Charter)
|
|
|
|
|
Delaware |
001-41949 |
81-3909017 |
(State or Other Jurisdiction
of Incorporation) |
(Commission File Number) |
(IRS Employer
Identification No.) |
|
|
|
|
|
5959 Horton Street 7th Floor |
|
Emeryville, California |
|
94608 |
(Address of Principal Executive Offices) |
|
(Zip Code) |
|
Registrant’s Telephone Number, Including Area Code: (510) 871-4880 |
(Former Name or Former Address, if Changed Since Last Report)
Check the appropriate box below if the Form 8-K filing is intended to simultaneously satisfy the filing obligation of the registrant under any of the following provisions:
☐Written communications pursuant to Rule 425 under the Securities Act (17 CFR 230.425)
☐Soliciting material pursuant to Rule 14a-12 under the Exchange Act (17 CFR 240.14a-12)
☐Pre-commencement communications pursuant to Rule 14d-2(b) under the Exchange Act (17 CFR 240.14d-2(b))
☐Pre-commencement communications pursuant to Rule 13e-4(c) under the Exchange Act (17 CFR 240.13e-4(c))
Securities registered pursuant to Section 12(b) of the Act:
|
|
|
|
|
Title of each class
|
|
Trading
Symbol(s) |
|
Name of each exchange on which registered
|
Common Stock, $0.0001 par value per share |
|
MGX |
|
Nasdaq Global Select Market |
Indicate by check mark whether the registrant is an emerging growth company as defined in Rule 405 of the Securities Act of 1933 (§ 230.405 of this chapter) or Rule 12b-2 of the Securities Exchange Act of 1934 (§ 240.12b-2 of this chapter).
Emerging growth company ☒
If an emerging growth company, indicate by check mark if the registrant has elected not to use the extended transition period for complying with any new or revised financial accounting standards provided pursuant to Section 13(a) of the Exchange Act. ☒
Item 2.02 Results of Operations and Financial Condition.
On May 11, 2026, Metagenomi Therapeutics, Inc. (the “Company”) announced its financial results for the quarter ended March 31, 2026 and additional business updates. A copy of the press release in connection with the announcement is being furnished as Exhibit 99.1 to this Current Report on Form 8-K and is incorporated herein by reference.
The information in this Item 2.02 (including Exhibit 99.1 attached hereto) is intended to be furnished and shall not be deemed “filed” for purposes of Section 18 of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), or otherwise subject to the liabilities of that section, nor shall it be deemed incorporated by reference in any filing under the Securities Act of 1933, as amended (the “Securities Act”), except as expressly set forth by specific reference in such filing.
Item 7.01 Regulation FD Disclosure.
A copy of the Company’s May 2026 corporate presentation is furnished as Exhibit 99.2 to this Current Report on Form 8-K and is incorporated herein by reference.
The information contained in this Item 7.01 (including Exhibit 99.2) is being furnished and shall not be deemed “filed” for purposes of Section 18 of the Exchange Act, or otherwise subject to the liabilities of that section and shall not be deemed incorporated by reference in any filing under the Securities Act or the Exchange Act, except as shall be expressly set forth by specific reference in such filing.
Item 9.01 Financial Statements and Exhibits.
(d) Exhibits
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned hereunto duly authorized.
|
|
|
|
|
|
|
Metagenomi Therapeutics, Inc. |
|
|
|
|
Date: |
May 11, 2026 |
By: |
/s/ Jian Irish |
|
|
|
Jian Irish, Ph.D., M.B.A.
President and Chief Executive Officer |
EX-99.1
Exhibit 99.1

Metagenomi Therapeutics Reports Business Updates and First Quarter 2026 Financial Results
On track for regulatory submission of MGX-001 to advance global clinical program, including investigational new drug application (“IND”) in 4Q 2026
Publication in Nature Structural & Molecular Biology highlights potential of MG119-28, a proprietary compact CRISPR nuclease with enhanced genome editing efficiency
$140.2 million in cash, cash equivalents, and available-for-sale marketable securities as of March 31, 2026, with runway anticipated to support operations through 4Q 2027
EMERYVILLE, Calif., May 11, 2026 (GLOBE NEWSWIRE) -- Metagenomi Therapeutics, Inc. (Nasdaq: MGX) (the “Company”), an in vivo genome editing company capitalizing on its proprietary technologies to create curative genetic medicines for patients, today reported financial results for the first quarter ended March 31, 2026, and provided business updates.
“We remain diligently focused on advancing our core genome-editing technologies, led by our MGX-001 program for hemophilia A, which remains on track for regulatory submission in the fourth quarter of this year and first-in-human studies in 2027,” said Jian Irish, Ph.D., M.B.A., President and Chief Executive Officer of Metagenomi Therapeutics. “The promise of our novel technology, most recently highlighted by a Nature publication, in addition to the encouraging preclinical data and continued IND-enabling execution, gives us confidence in our goal to provide patients an option for one-time, curative treatments, beginning with hemophilia A.”
First Quarter 2026 and Subsequent Updates
MGX-001 – Hemophilia A Program
•On track for regulatory submission of MGX-001 to advance global clinical program, including an IND in the fourth quarter of 2026, and subject to regulatory clearance, initiate clinical trials in 2027.
•During the first quarter, Kapil Saxena, MD joined the Company to spearhead the clinical development program for MGX-001. Prior to joining the Company, Dr. Saxena held leadership positions in clinical development at Autolus, Daiichi Sankyo and Bayer. Prior to joining industry, Dr. Saxena was a practicing hematologist and director of hemophilia treatment centers in Boston and Oklahoma.
MGX-001 Large Gene Integration System for Protein Replacement via Gene Insertion
•Following the demonstration of in vivo proof-of-concept in NHPs via the MGX-001 site-specific genome integration system, the Company is pursuing disease indications which have the potential to be treated by protein replacement via gene insertion.
Platform Technology Updates
•Publication in Nature Structural & Molecular Biology highlights the discovery and detailed characterization of MG119-28, a compact CRISPR nuclease with superior editing efficiency relative to previously identified compact nucleases from the Cas12f class.
First Quarter 2026 Financial Results
Cash Position: Cash, cash equivalents, and available-for-sale marketable securities were $140.2 million as of March 31, 2026.
R&D Expenses: Research and development (R&D) expenses were $19.3 million for the quarter ended March 31, 2026, compared to $25.1 million for the comparable period in 2025.
G&A Expenses: General and administrative (G&A) expenses were $6.5 million for the quarter ended March 31, 2026, compared to $6.8 million for the comparable period in 2025.
About Metagenomi Therapeutics
Metagenomi Therapeutics, Inc. is an in vivo genome editing company capitalizing on its proprietary technologies to create curative genetic medicines for patients. The Company was founded on the science of metagenomics, the study of genetic materials recovered from the natural environment, to discover and develop a suite of novel CRISPR gene-editing tools potentially capable of correcting any type of genetic mutation found anywhere in the human genome. The Company focuses on high value programs in disease indications with well-understood biology and clearly defined clinical development and regulatory pathways. Going forward, the Company intends to continue to expand its pipeline by leveraging its proprietary genetic editing capabilities in site specific deletion, insertion and correction.
MGX-001, the Company’s lead, wholly-owned development program in hemophilia A, has demonstrated a preclinical profile with best-in-class treatment potential, including targeted genome editing and durable gene expression in a one-time treatment. MGX-001 is designed to provide curative, life-long protection from bleeding events and joint damage in adults and children, potentially enabling a new standard of care for the treatment of hemophilia A. The Company is also currently pursuing indications leveraging the MGX-001 site-specific genome integration system and partnered assets targeting cardiometabolic diseases. For more information, please visit https://metagenomi.co/.
Cautionary Note Regarding Forward‐Looking Statements
This press release contains “forward-looking statements” within the meaning of Section 27A of the Securities Act of 1933 and Section 21E of the Securities Exchange Act of 1934, each as amended. Such statements, which are often indicated by terms such as “anticipate,” “believe,” “could,” “estimate,” “expect,” “goal,” “intend,” “look forward to,” “may,” “plan,” “potential,” “predict,” “project,” “should,” “will,” “would” and similar expressions include, but are not limited to, any statements relating to our product development programs, including the timing of and our ability to conduct IND-enabling studies and make regulatory filings such as INDs, expectations
regarding MGX-001 including the preclinical profile with best-in-class treatment potential and timing to submit the IND/CTA package, statements regarding the Company’s plans to prioritize its preclinical pipeline and potential for value creation and sustainable growth, statements regarding upcoming milestones, statements concerning the potential of therapies and product candidates, statements concerning the impact of the organizational restructuring, statements concerning our anticipated cash runway, and any other statements that are not historical facts. Forward-looking statements are based on management’s current expectations and are subject to risks and uncertainties that could negatively affect our business, operating results, financial condition, and stock value. Factors that could cause actual results to differ materially from those currently anticipated include: risks relating to our growth strategy; our ability to obtain, perform under, and maintain financing and strategic agreements and relationships; risks relating to the results of research and development activities; risks relating to the timing of IND submissions and starting and completing clinical trials; uncertainties relating to preclinical and clinical testing; our dependence on third party suppliers; our ability to attract, integrate and retain key personnel; the early stage of products under development; our need for substantial additional funds; government regulation and the current regulatory environment; patent and intellectual property matters; competition; the volatility of capital markets and other adverse macroeconomic factors; as well as other risks described in “Risk Factors,” in our most recent Form 10-K and other risk factors set forth from time to time in our filings with the Securities and Exchange Commission made pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934, as amended. We expressly disclaim any obligation or undertaking to release publicly any updates or revisions to any forward-looking statements contained herein to reflect any change in our expectations or any changes in events, conditions or circumstances on which any such statement is based, except as required by law, and we claim the protection of the safe harbor for forward-looking statements contained in the Private Securities Litigation Reform Act of 1995.
Investor Contact:
Stephen Jasper
Gilmartin Group
stephen@gilmartinir.com
Condensed Financial Statements
Condensed Balance Sheet Data
(Unaudited)
|
|
|
|
|
|
|
|
|
|
|
March 31, |
|
|
December 31, |
|
(in thousands) |
|
2026 |
|
|
2025 |
|
Cash, cash equivalents and available-for-sale marketable securities |
|
$ |
140,162 |
|
|
$ |
160,799 |
|
Total assets |
|
$ |
196,953 |
|
|
$ |
221,103 |
|
Total liabilities |
|
$ |
59,168 |
|
|
$ |
62,507 |
|
Total stockholders’ equity |
|
$ |
137,785 |
|
|
$ |
158,596 |
|
Total liabilities and stockholders’ equity |
|
$ |
196,953 |
|
|
$ |
221,103 |
|
Condensed Statements of Operations
(Unaudited)
|
|
|
|
|
|
|
|
|
|
|
Three Months Ended
March 31, |
|
(In thousands, except share and per share data) |
|
2026 |
|
|
2025 |
|
Collaboration revenue |
|
$ |
1,248 |
|
|
$ |
4,127 |
|
Operating expenses: |
|
|
|
|
|
|
Research and development |
|
|
19,300 |
|
|
|
25,142 |
|
General and administrative |
|
|
6,535 |
|
|
|
6,805 |
|
Total operating expenses |
|
|
25,835 |
|
|
|
31,947 |
|
Loss from operations |
|
|
(24,587 |
) |
|
|
(27,820 |
) |
Other income (expense): |
|
|
|
|
|
|
Interest income |
|
|
1,539 |
|
|
|
2,887 |
|
Other expense, net |
|
|
(1 |
) |
|
|
(8 |
) |
Total other income, net |
|
|
1,538 |
|
|
|
2,879 |
|
Net loss before provision for income taxes |
|
|
(23,049 |
) |
|
|
(24,941 |
) |
Provision for income taxes |
|
|
(10 |
) |
|
|
(98 |
) |
Net loss |
|
$ |
(23,059 |
) |
|
$ |
(25,039 |
) |
Net loss per share attributable to common stockholders, basic and diluted |
|
$ |
(0.61 |
) |
|
$ |
(0.68 |
) |
Weighted average common shares outstanding, basic and diluted |
|
|
37,581,094 |
|
|
|
37,019,027 |
|

Precision gene editing designed to deliver durable, curative medicines��Hemophilia A lead program | IND 2026 Corporate Presentation May 2026

This presentation includes forward-looking statements, including forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995. All statements other than statements of historical facts contained in this presentation are forward looking statements, including statements regarding our cash runway, strategy and plans, industry environment, potential growth opportunities, and the therapeutic potential of our programs. The words “believe,” “may,” “will,” “estimate,” “continue,” “anticipate,” “design,” “expect,” “could,” “plan,” “potential,” “predict,” “seek,” “should,” “would,” or the negative version of these words and similar expressions are intended to identify forward-looking statements. We have based these forward-looking statements on our current expectations and projections about future events and trends that we believe may affect our financial condition, results of operations, strategy, short- and long-term business operations and objectives, and financial needs. These forward-looking statements are subject to a number of risks, uncertainties and assumptions, including but not limited to, our ability to develop and advance our programs and product candidates, our ability to maintain and establish collaborations or strategic partnerships, our regulatory approvals and filings, and other risks, uncertainties and assumptions identified in our filings with the Securities and Exchange Commission (the “SEC”), including our most recent Form 10-K and Form 10-Q filed with the SEC, and any subsequent filings with the SEC. Moreover, we operate in a very competitive and rapidly changing environment, and it is not possible for our management to predict all risks, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in any forward-looking statements we may make. In light of these risks, uncertainties and assumptions, the forward-looking statements and circumstances discussed in this presentation may not occur and actual results could differ materially and adversely from those anticipated or implied in the forward-looking statements. You should not rely upon forward-looking statements as predictions of future events. Although we believe that the expectations reflected in the forward-looking statements are reasonable, we cannot guarantee that the future results, levels of activity, performance or events and circumstances reflected in the forward-looking statements will be achieved or occur. Moreover, except as required by law, neither we nor any other person assumes responsibility for the accuracy and completeness of the forward-looking statements. We undertake no obligation to update publicly any forward-looking statements for any reason after the date of this presentation to conform these statements to actual results or to changes in our expectations, unless required by law. This presentation contains estimates and other information concerning our industry, our business and the markets for our products. Information that is based on estimates, market research or similar methodologies is inherently subject to uncertainties, and actual events or circumstances may differ materially from events and circumstances that are assumed in this information. Unless otherwise expressly stated, we obtained this industry, business, market and other data from our own internal estimates and research as well as from reports, research surveys, studies and similar data prepared by market research firms and other third parties, industry, medical and general publications, government data and similar sources. These sources include government and industry sources. Industry publications and surveys generally state that the information contained therein has been obtained from sources believed to be reliable. Although we believe the industry and market data to be reliable as of the date of this presentation, this information could prove to be inaccurate. Industry and market data could be wrong because of the method by which sources obtained their data and because information cannot always be verified with complete certainty due to the limits on the availability and reliability of raw data, the voluntary nature of the data gathering process and other limitations and uncertainties. While we believe our internal company estimates and research as to such matters is reliable and the market definitions are appropriate, neither such research nor these definitions have been verified by any independent source, and no reliance should be placed on or should be made on any information or statements made in this presentation relating to or based on such internal estimates and research. Forward-looking statements

A differentiated in vivo gene editing company advancing curative genetic medicines An in vivo CRISPR gene editing company capitalizing on its proprietary technologies to create curative genetic medicines Focusing on lead program MGX-001 in hemophilia A advancing to the clinic Expanding indications leveraging site-specific gene integration system and partnered assets targeting cardiometabolic indications
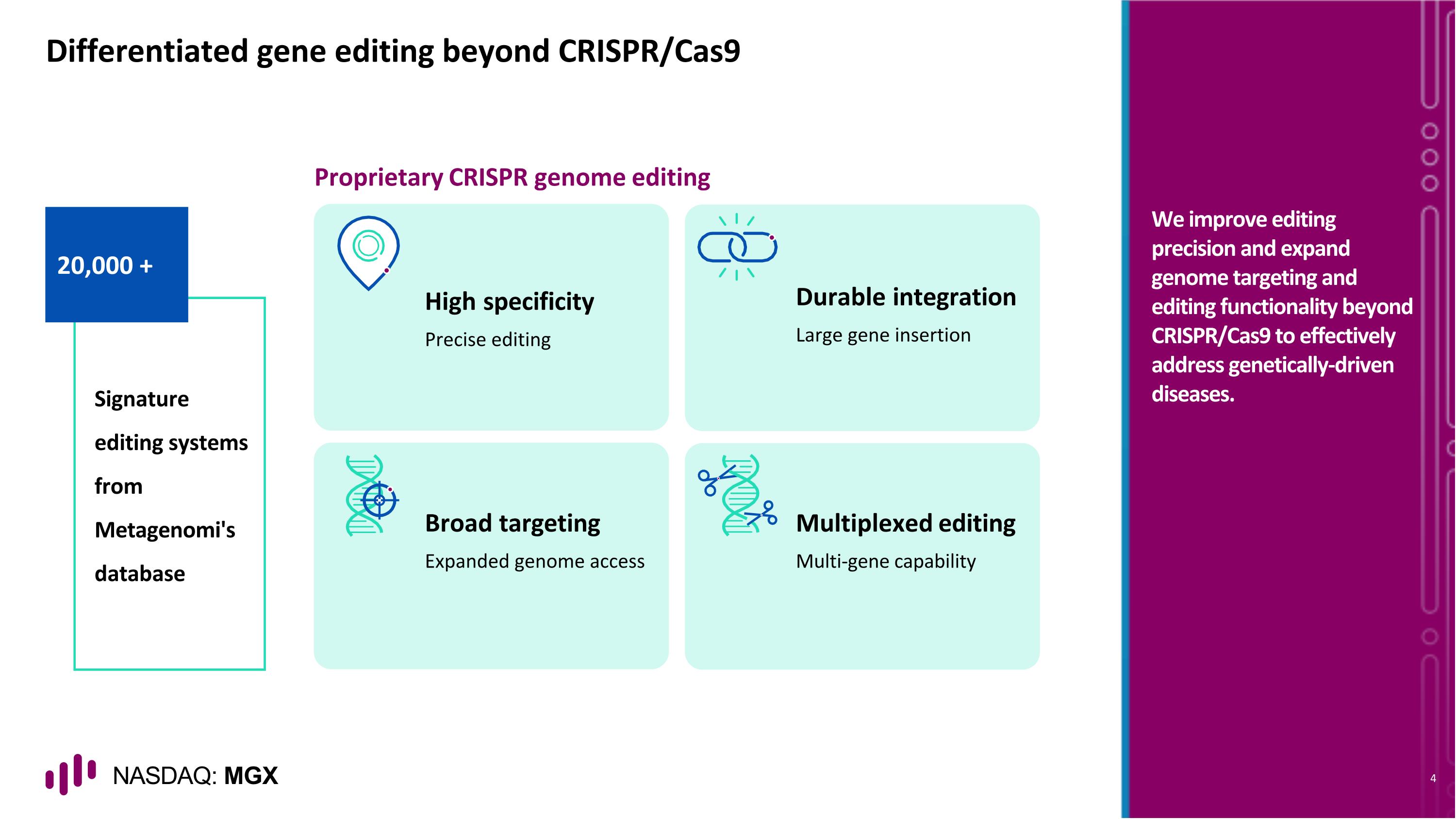
20,000 + Differentiated gene editing beyond CRISPR/Cas9 Signature editing systems from Metagenomi's database High specificity Precise editing Multiplexed editing Multi-gene capability Durable integration Large gene insertion Broad targeting Expanded genome access Proprietary CRISPR genome editing We improve editing precision and expand genome targeting and editing functionality beyond CRISPR/Cas9 to effectively address genetically-driven diseases.
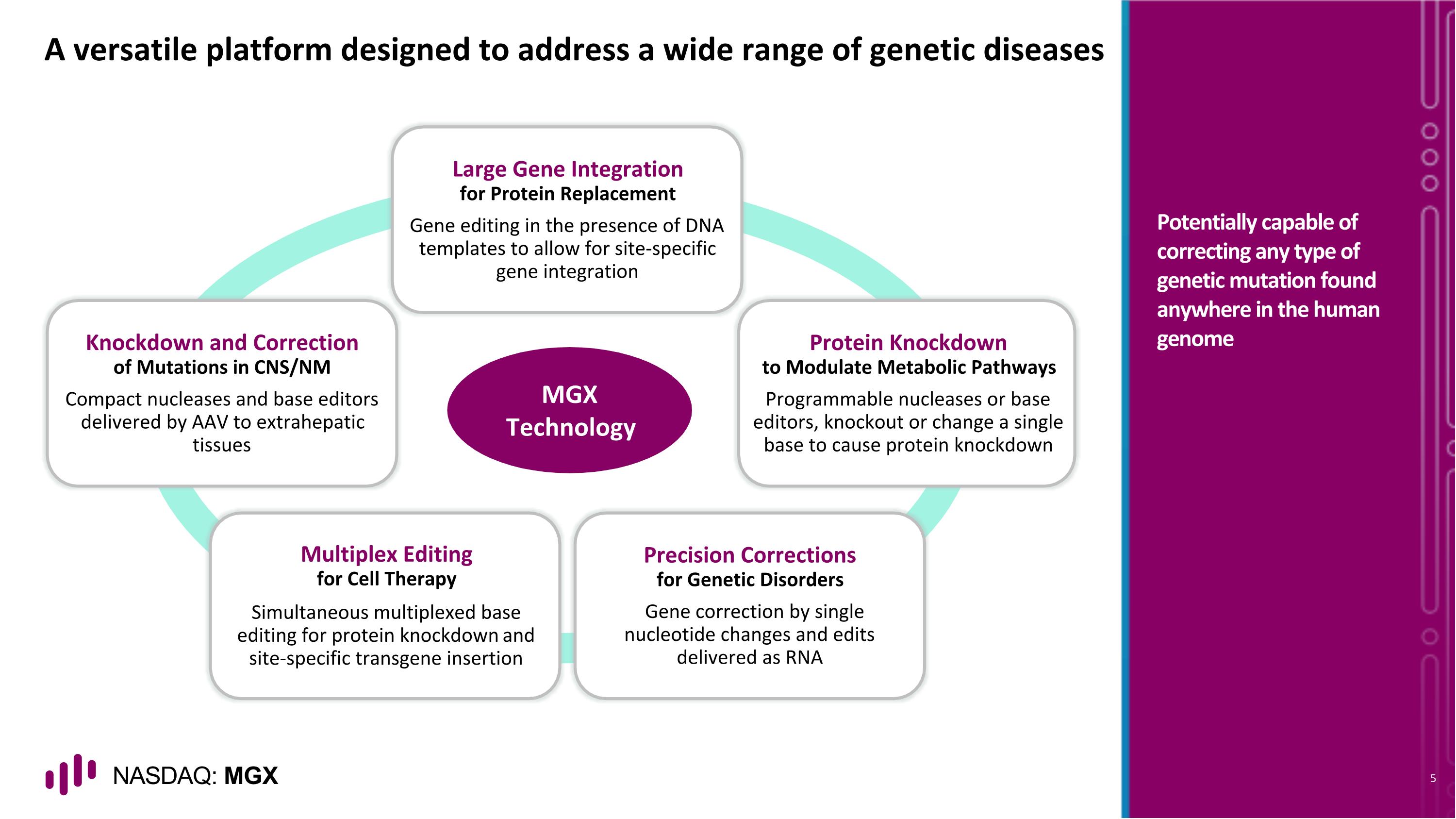
A versatile platform designed to address a wide range of genetic diseases Potentially capable of correcting any type of genetic mutation found anywhere in the human genome Large Gene Integration for Protein Replacement Gene editing in the presence of DNA templates to allow for site-specific gene integration Protein Knockdown to Modulate Metabolic Pathways Programmable nucleases or base editors, knockout or change a single base to cause protein knockdown Precision Corrections for Genetic Disorders Gene correction by single nucleotide changes and edits delivered as RNA Multiplex Editing for Cell Therapy Simultaneous multiplexed base editing for protein knockdown and site-specific transgene insertion Knockdown and Correction of Mutations in CNS/NM Compact nucleases and base editors delivered by AAV to extrahepatic tissues MGX Technology
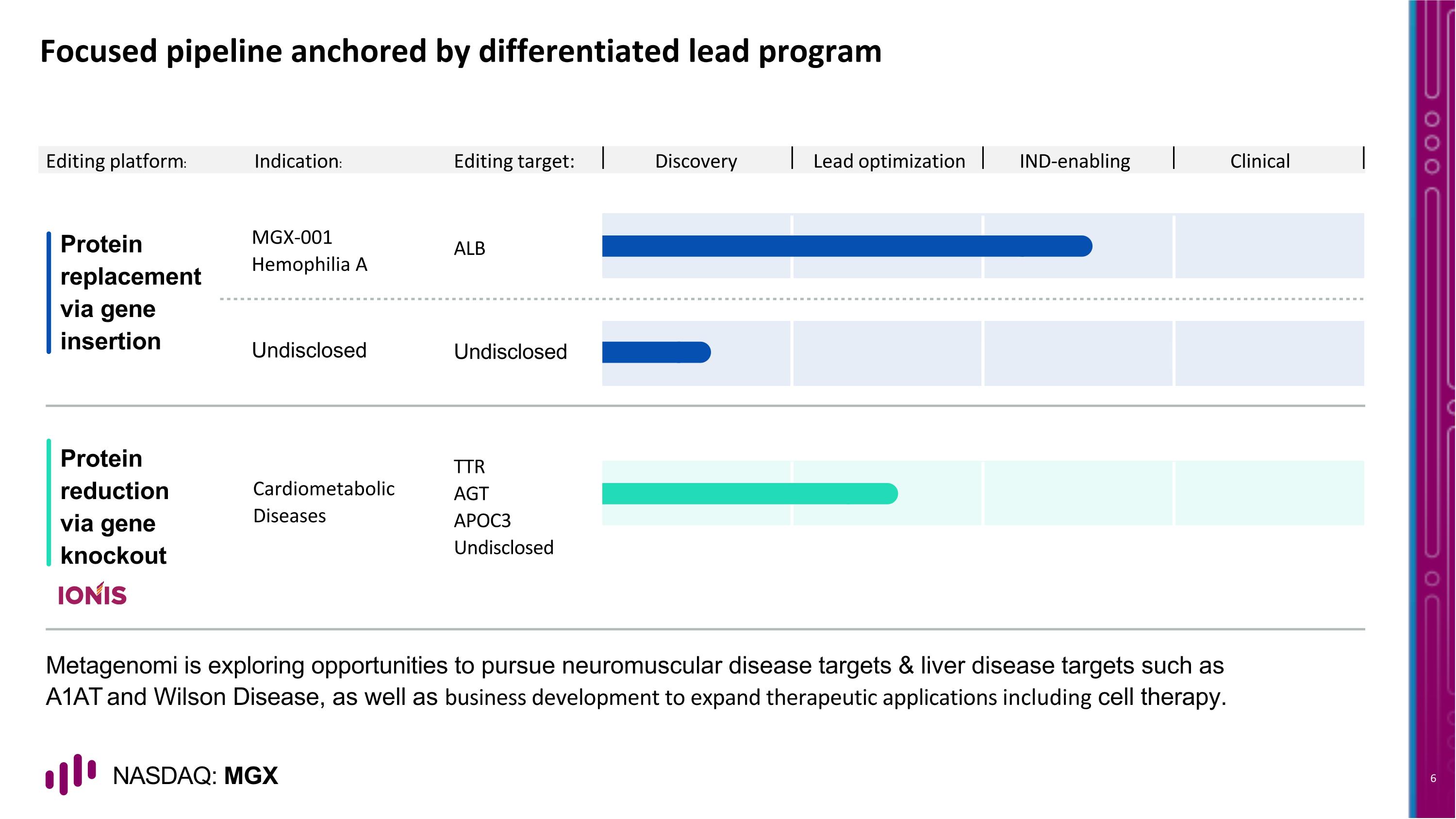
Focused pipeline anchored by differentiated lead program Protein replacement via gene insertion Protein reduction via gene knockout ALB MGX-001 Hemophilia A Editing platform: Editing target: Discovery Lead optimization IND-enabling Clinical Undisclosed Undisclosed TTR AGT APOC3 Undisclosed Cardiometabolic Diseases Indication: Metagenomi is exploring opportunities to pursue neuromuscular disease targets & liver disease targets such as A1AT and Wilson Disease, as well as business development to expand therapeutic applications including cell therapy.
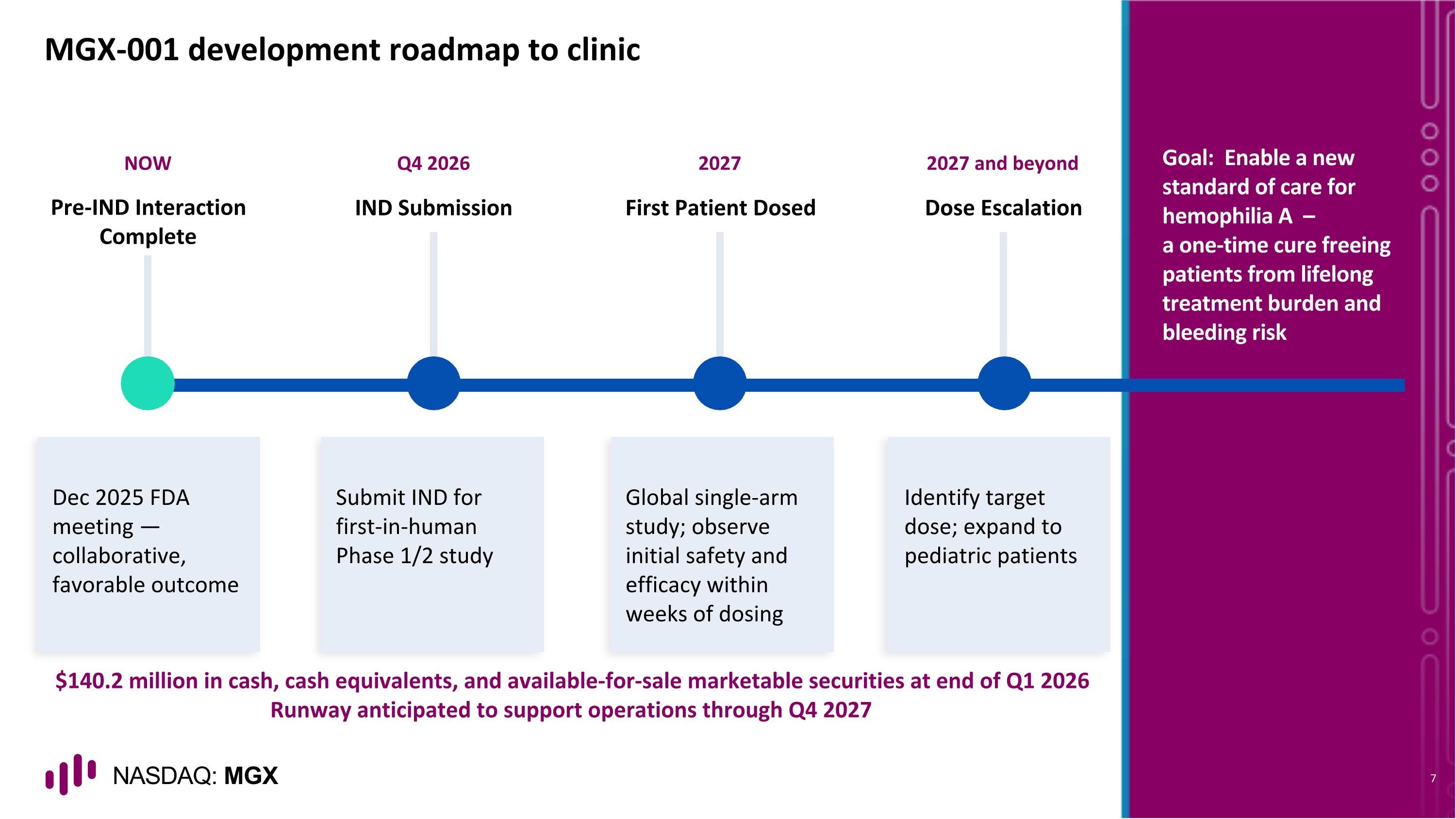
NOW Pre-IND Interaction Complete Dec 2025 FDA meeting — collaborative, favorable outcome Q4 2026 IND Submission Submit IND for first-in-human Phase 1/2 study 2027 First Patient Dosed Global single-arm study; observe initial safety and efficacy within weeks of dosing 2027 and beyond Dose Escalation Identify target dose; expand to pediatric patients MGX-001 development roadmap to clinic Goal: Enable a new standard of care for hemophilia A – a one-time cure freeing patients from lifelong treatment burden and bleeding risk $140.2 million in cash, cash equivalents, and available-for-sale marketable securities at end of Q1 2026 Runway anticipated to support operations through Q4 2027

MGX-001 - designed to deliver a durable, one-time treatment for Hemophilia A

Hemophilia A: large, validated market still lacking a durable cure Hemophilia A is the most common X-linked inherited and de novo bleeding disorder, largely affecting males. Caused by variety of mutations in the Factor VIII (FVIII) gene leading to loss of functional FVIII protein. Current Standard of Care: ~26,500 patients in the U.S.1 ~500,000 worldwide2 - Soucie, J.M., et al, 2020. Haemophilia. Vol. 26, no. 3, pp. 487–493. - Stonebraker, J. S., et al, 2010. Haemophilia. Vol. 16, pp. 20–32. - ICER. Gene Therapy for Hemophilia B and A: Final Evidence Report. Dec 22, 2022. - Curtis R et al. Poster presented at: 65th ASH Annual Meeting & Exposition; December 11, 2023; San Diego, CA. Factor VIII replacement therapy IV typically dosed 1 - 3 times/week Significant adherence challenges Risk of breakthrough bleeding Chronic treatment, non-curative Bi-specific antibody "mimetic" SQ dosed 1, 2 or 4 weeks post loading Risk of breakthrough bleeding Treatment burden, non-curative Variable initial efficacy Decline in FVIII levels over time High risk of prolonged corticosteroid use Not suitable for pediatric patients Annual treatment cost3: ~$565K - $750K Lifetime treatment cost: ~$18M - $24M4 FVIII Recent Curative Gene Therapy Attempted:
Strong advocacy and infrastructure Robust preclinical models and regulatory familiarity Clearly defined target threshold of curative FVIII level & wide safety range Monogenic and well-characterized biology with clear biomarker Genome editing offers a potentially ideal curative approach for Hemophilia A proprietary Type V nuclease IND-enabling stage Technology: Durability: Regulatory status: Pediatric potential: Hemophilia A is an ideal indication for genome editing approach: MGX-001 is uniquely suited for patients of all ages: MGX-001 is a potentially durable, curative approach for adults and children – the population with the most to gain Clear opportunity for a durable cure

Compelling preclinical profile achieved across efficacy, durability, and safety Extensive and supportive preclinical data set FVIII activity achieved in curative range with clear dose response in NHPs Durable FVIII activity over approximately 19-month study in NHPs Encouraging safety profile with single doses of steroids, and no genotoxicity observed Novel mechanism of action FVIII integration leveraging albumin promoter to achieve normalized activity level Promoterless FVIII gene delivered by AAV effective at lower dose than approved gene therapies Precise FVIII integration facilitated by proprietary CRISPR nuclease MG29-1 achieving no detectable off-target editing Compelling potential clinical profile Enables endogenous production of FVIII supporting hemostatic regulation Potential to normalize FVIII levels and deliver meaningful clinical benefit for both adults and pediatric patients Goal to be one-time durable cure allowing patients the freedom of a hemophilia free mind
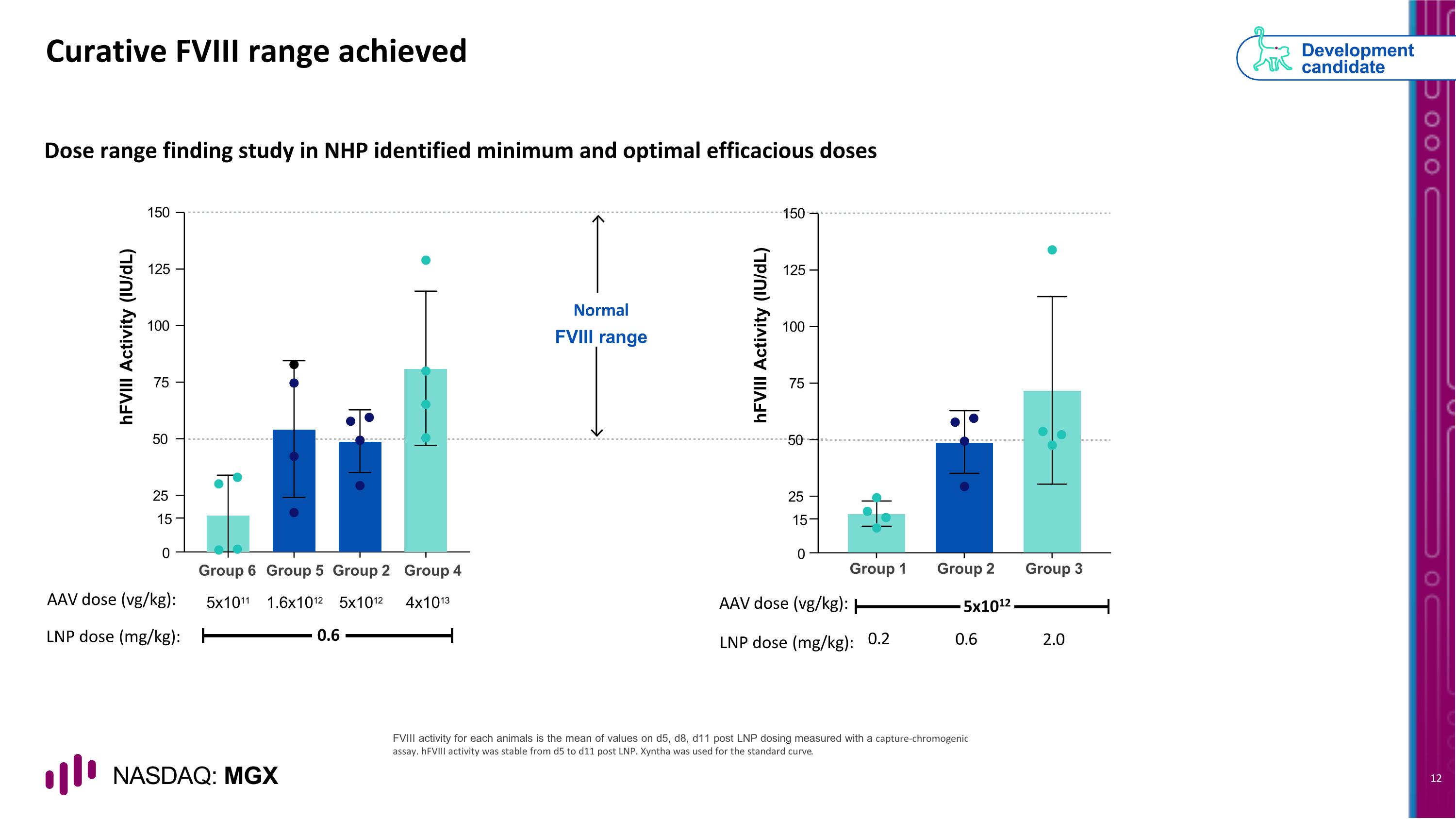
Curative FVIII range achieved Dose range finding study in NHP identified minimum and optimal efficacious doses Normal FVIII range FVIII activity for each animals is the mean of values on d5, d8, d11 post LNP dosing measured with a capture-chromogenic assay. hFVIII activity was stable from d5 to d11 post LNP. Xyntha was used for the standard curve. Development candidate 25 15 0 50 75 100 125 150 hFVIII Activity (IU/dL) Group 6 Group 5 Group 2 Group 4 AAV dose (vg/kg): 5x1011 1.6x1012 5x1012 4x1013 LNP dose (mg/kg): 0.6 hFVIII Activity (IU/dL) 25 15 0 50 75 100 125 150 Group 2 Group 3 5x1012 0.6 2.0 Group 1 0.2 AAV dose (vg/kg): LNP dose (mg/kg):
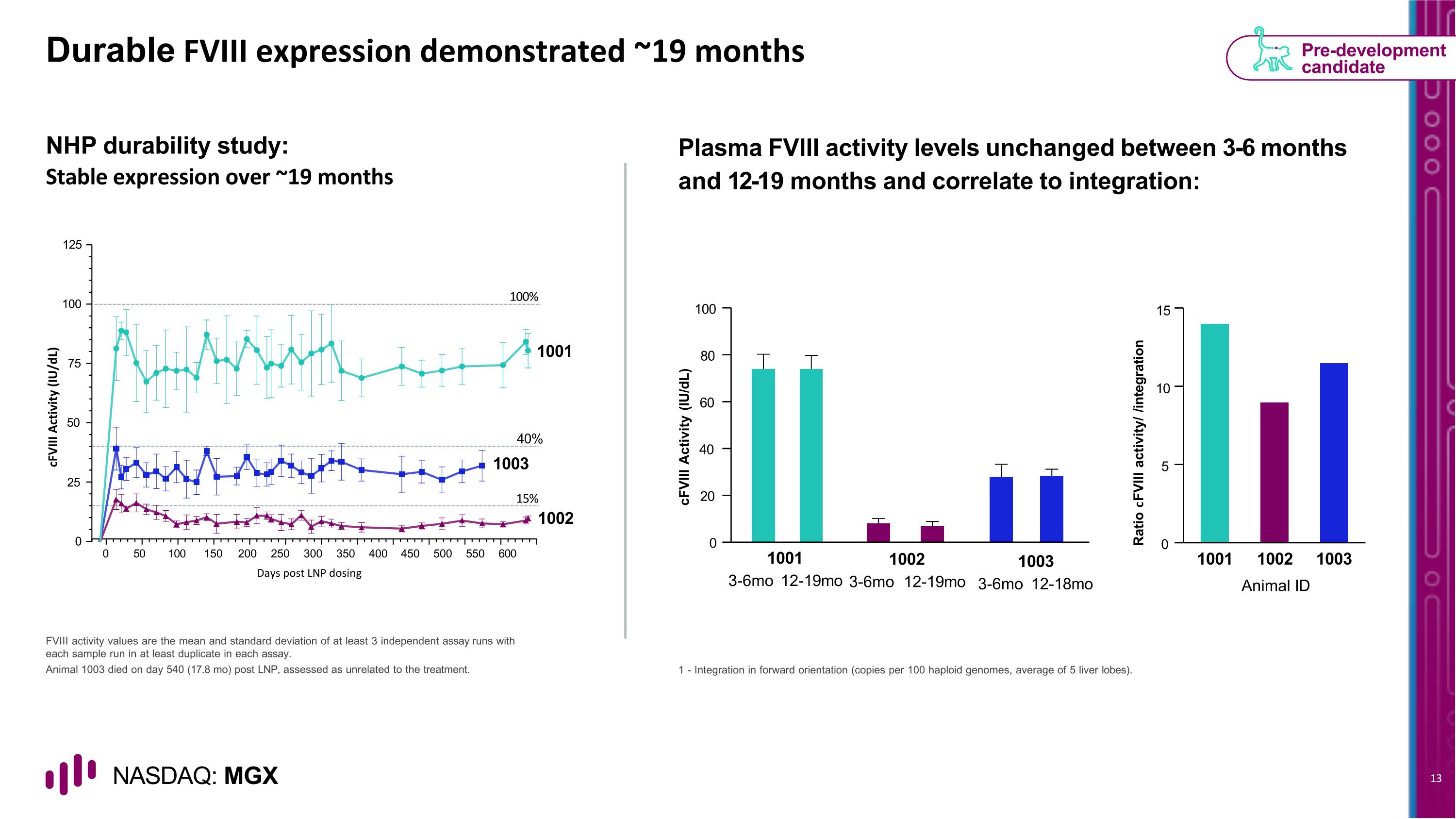
Durable FVIII expression demonstrated ~19 months NHP durability study: Stable expression over ~19 months Plasma FVIII activity levels unchanged between 3-6 months and 12-19 months and correlate to integration: FVIII activity values are the mean and standard deviation of at least 3 independent assay runs with each sample run in at least duplicate in each assay. Animal 1003 died on day 540 (17.8 mo) post LNP, assessed as unrelated to the treatment. 1 - Integration in forward orientation (copies per 100 haploid genomes, average of 5 liver lobes). Pre-development candidate 0 20 40 60 80 100 cFVIII Activity (IU/dL) 1001 3-6mo 12-19mo 1002 3-6mo 12-19mo 1003 3-6mo 12-18mo Ratio cFVIII activity/ /integration Animal ID 0 5 10 15 1001 1002 1003 0 50 100 150 200 250 300 350 400 450 500 550 600 100 75 50 25 0 125 1001 1003 40% 15% 1002 cFVIII Activity (IU/dL) Days post LNP dosing 100%
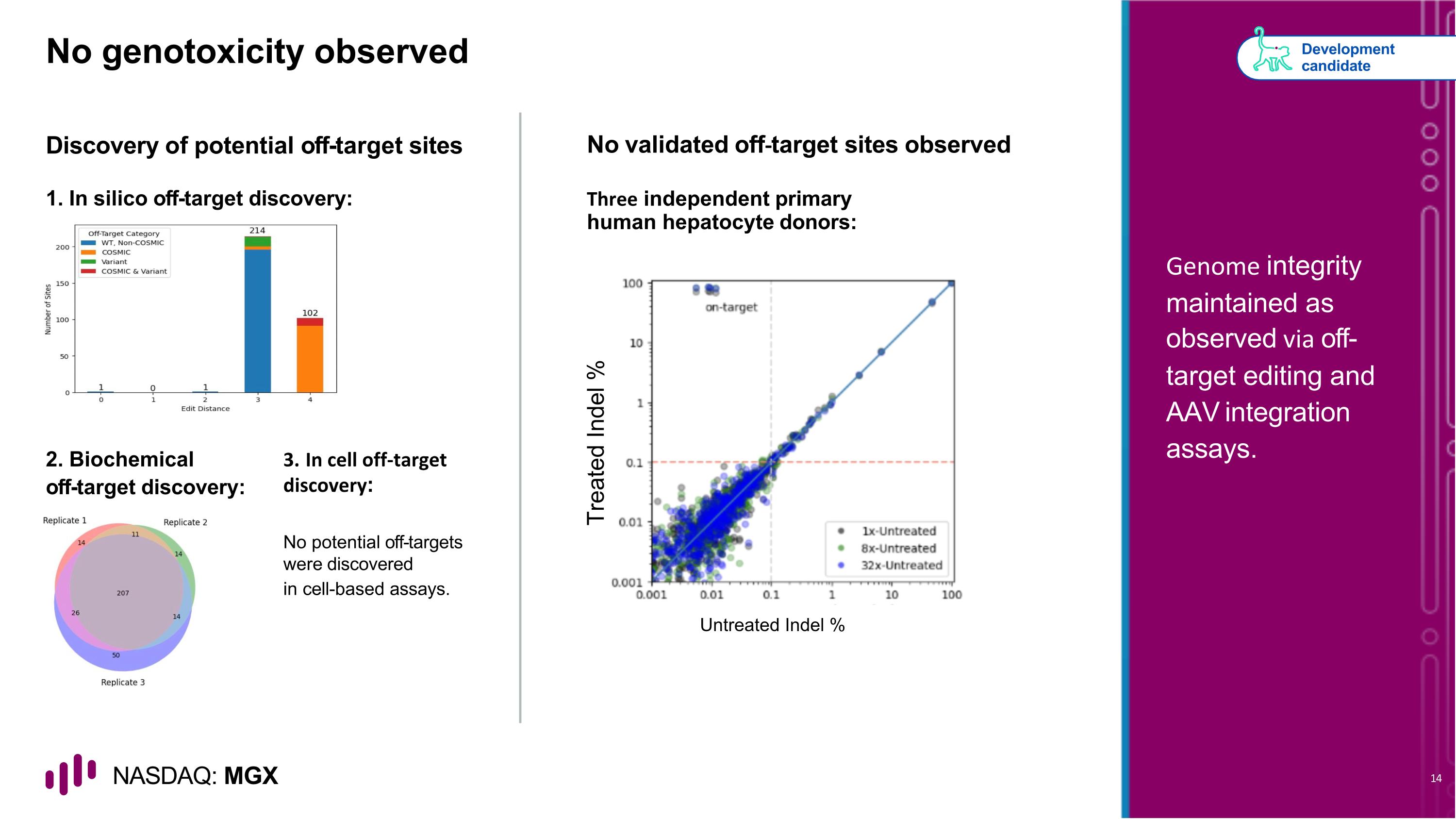
No genotoxicity observed Discovery of potential off-target sites No validated off-target sites observed 2. Biochemical off-target discovery: No potential off-targets were discovered in cell-based assays. Development candidate Genome integrity maintained as observed via off-target editing and AAV integration assays. 1. In silico off-target discovery: Three independent primary human hepatocyte donors: Untreated Indel % Treated Indel % 3. In cell off-target discovery:
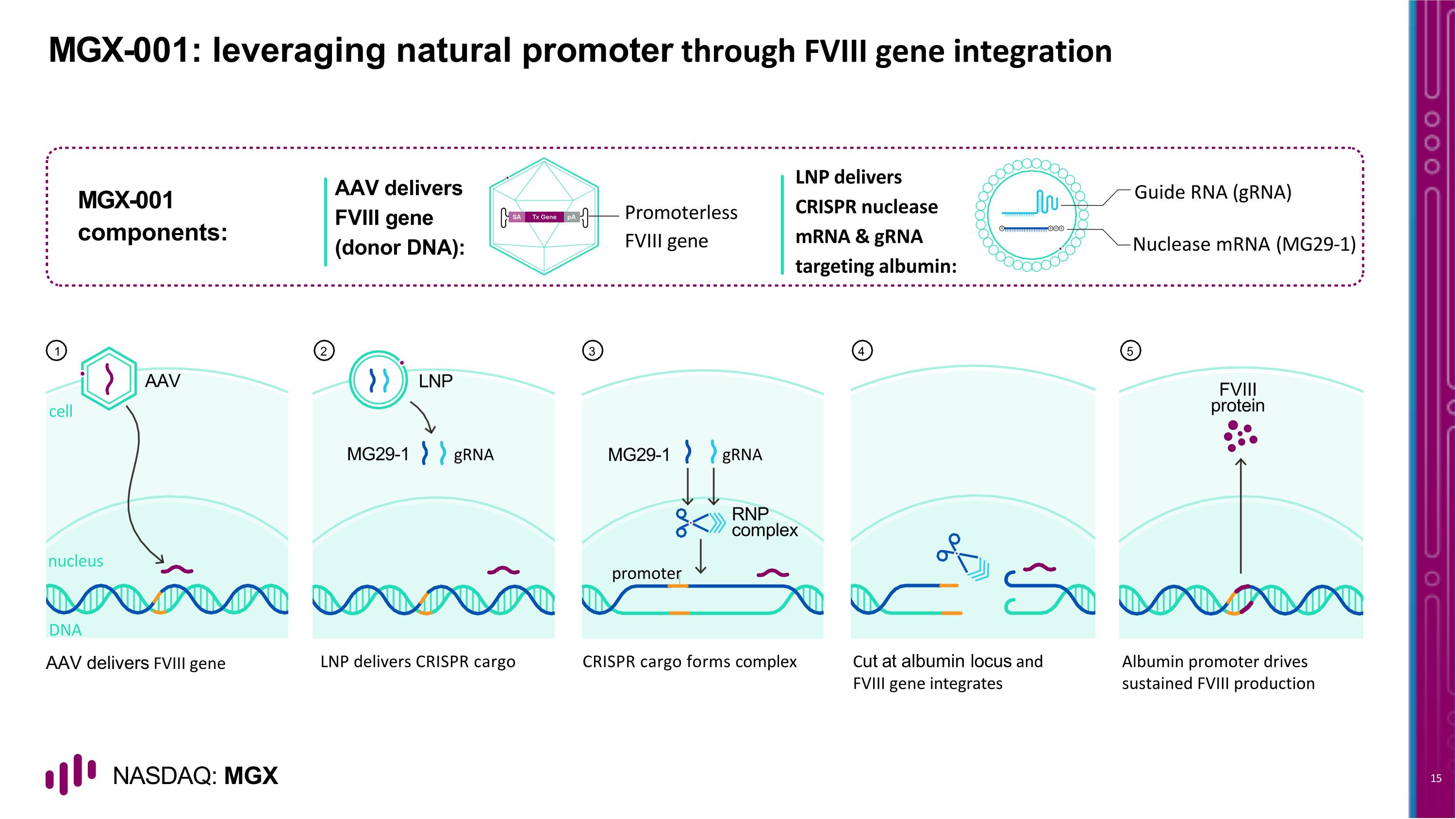
MGX-001: leveraging natural promoter through FVIII gene integration AAV delivers FVIII gene LNP delivers CRISPR cargo CRISPR cargo forms complex Cut at albumin locus and FVIII gene integrates Albumin promoter drives sustained FVIII production AAV delivers FVIII gene (donor DNA): Promoterless FVIII gene MGX-001 components: Guide RNA (gRNA) Nuclease mRNA (MG29-1) LNP delivers CRISPR nuclease mRNA & gRNA targeting albumin: 1 2 3 4 5 AAV cell nucleus DNA LNP gRNA MG29-1 FVIII protein gRNA promoter MG29-1 RNP complex

Our goal: To enable a new standard of care for hemophilia A Potential to deliver a durable cure for both adult and pediatric patients with hemophilia A Designed to enable endogenous FVIII expression for hemostatic regulation Compelling pre-clinical data Curative FVIII activity Durable FVIII expression Encouraging safety profile Minimal steroid use Promoterless AAV application No off-target editing observed Established regulatory framework and defined clinical endpoints pre-IND interaction completed IND submission on track for Q4 2026 First-in human in 2027

Expanding applications of site-specific large gene integration system
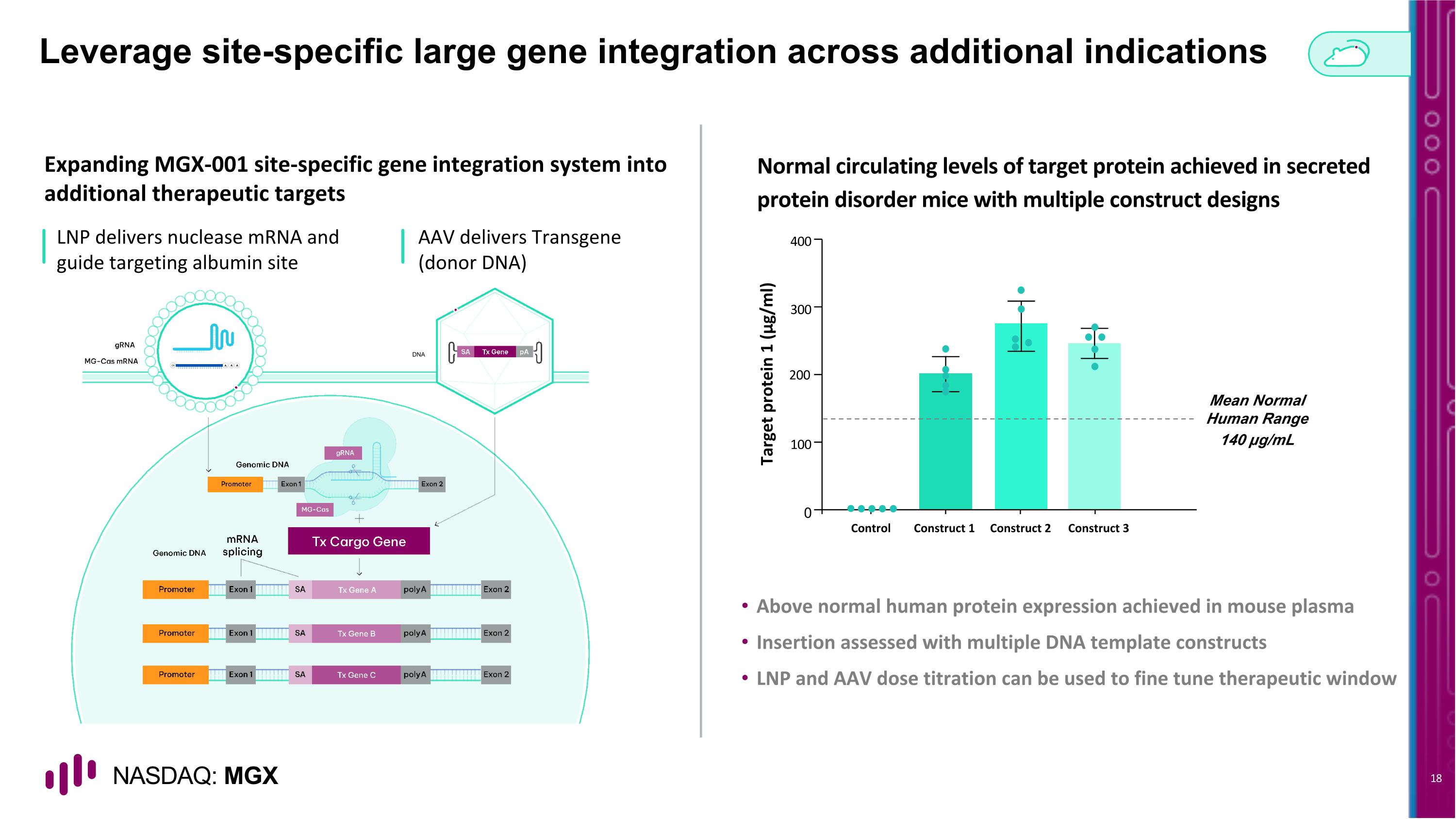
Leverage site-specific large gene integration across additional indications Expanding MGX-001 site-specific gene integration system into additional therapeutic targets Normal circulating levels of target protein achieved in secreted protein disorder mice with multiple construct designs LNP delivers nuclease mRNA and guide targeting albumin site Above normal human protein expression achieved in mouse plasma Insertion assessed with multiple DNA template constructs LNP and AAV dose titration can be used to fine tune therapeutic window Mean Normal Human Range 140 µg/mL Target protein 1 (μg/ml) 0 100 200 300 400 Construct 1 Construct 2 Control Construct 3 AAV delivers Transgene�(donor DNA) DNA
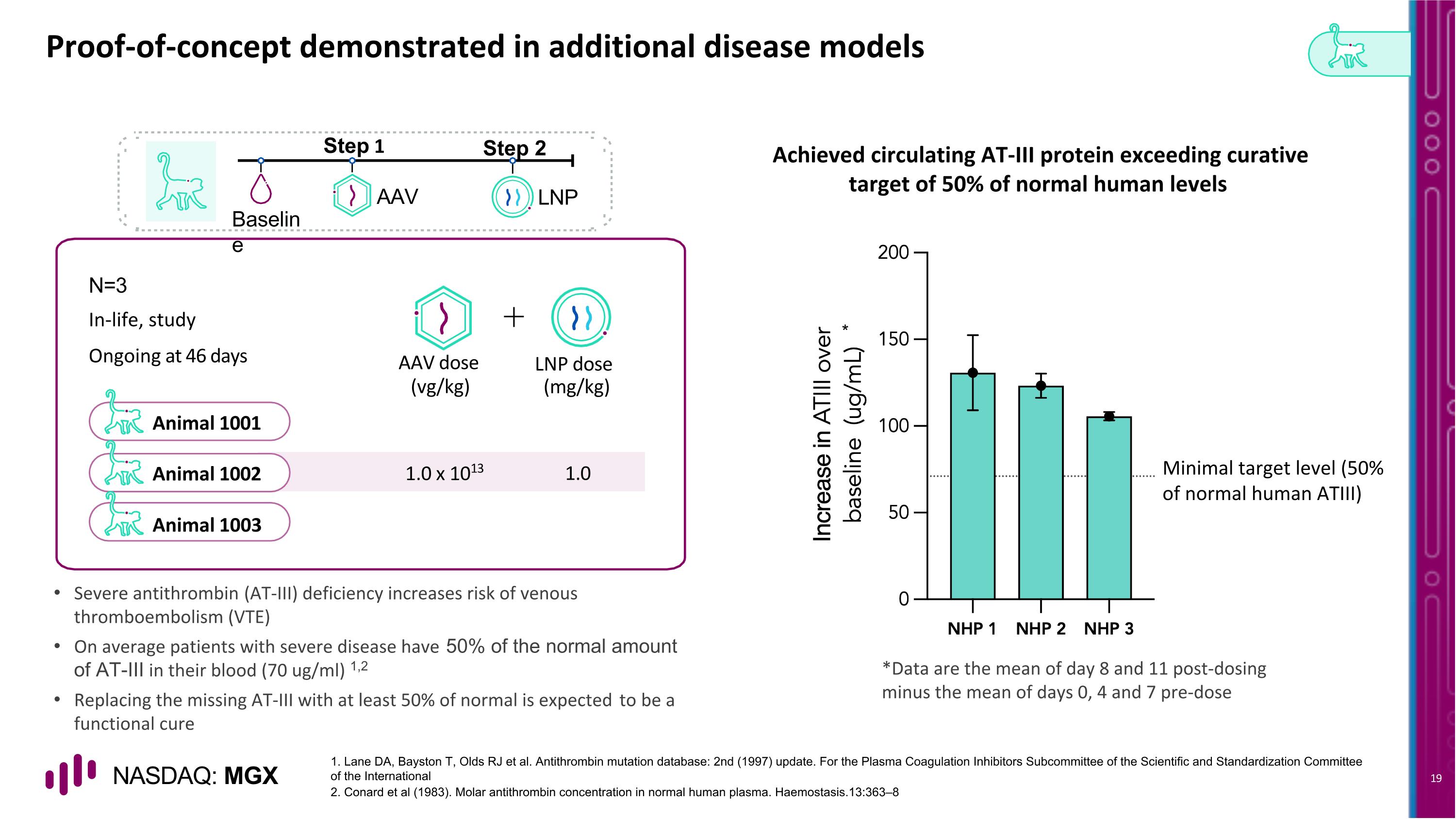
Animal 1001 Animal 1002 Animal 1003 Proof-of-concept demonstrated in additional disease models N=3 AAV LNP Baseline Step 1 Step 2 In-life, study Ongoing at 46 days AAV dose LNP dose (vg/kg) (mg/kg) 1.0 x 1013 1.0 1. Lane DA, Bayston T, Olds RJ et al. Antithrombin mutation database: 2nd (1997) update. For the Plasma Coagulation Inhibitors Subcommittee of the Scientific and Standardization Committee of the International 2. Conard et al (1983). Molar antithrombin concentration in normal human plasma. Haemostasis.13:363–8 Achieved circulating AT-III protein exceeding curative target of 50% of normal human levels Severe antithrombin (AT-III) deficiency increases risk of venous thromboembolism (VTE) On average patients with severe disease have 50% of the normal amount of AT-III in their blood (70 ug/ml) 1,2 Replacing the missing AT-III with at least 50% of normal is expected to be a functional cure Minimal target level (50% of normal human ATIII) * *Data are the mean of day 8 and 11 post-dosing minus the mean of days 0, 4 and 7 pre-dose

Broaden therapeutic potential with transformative gene editing�
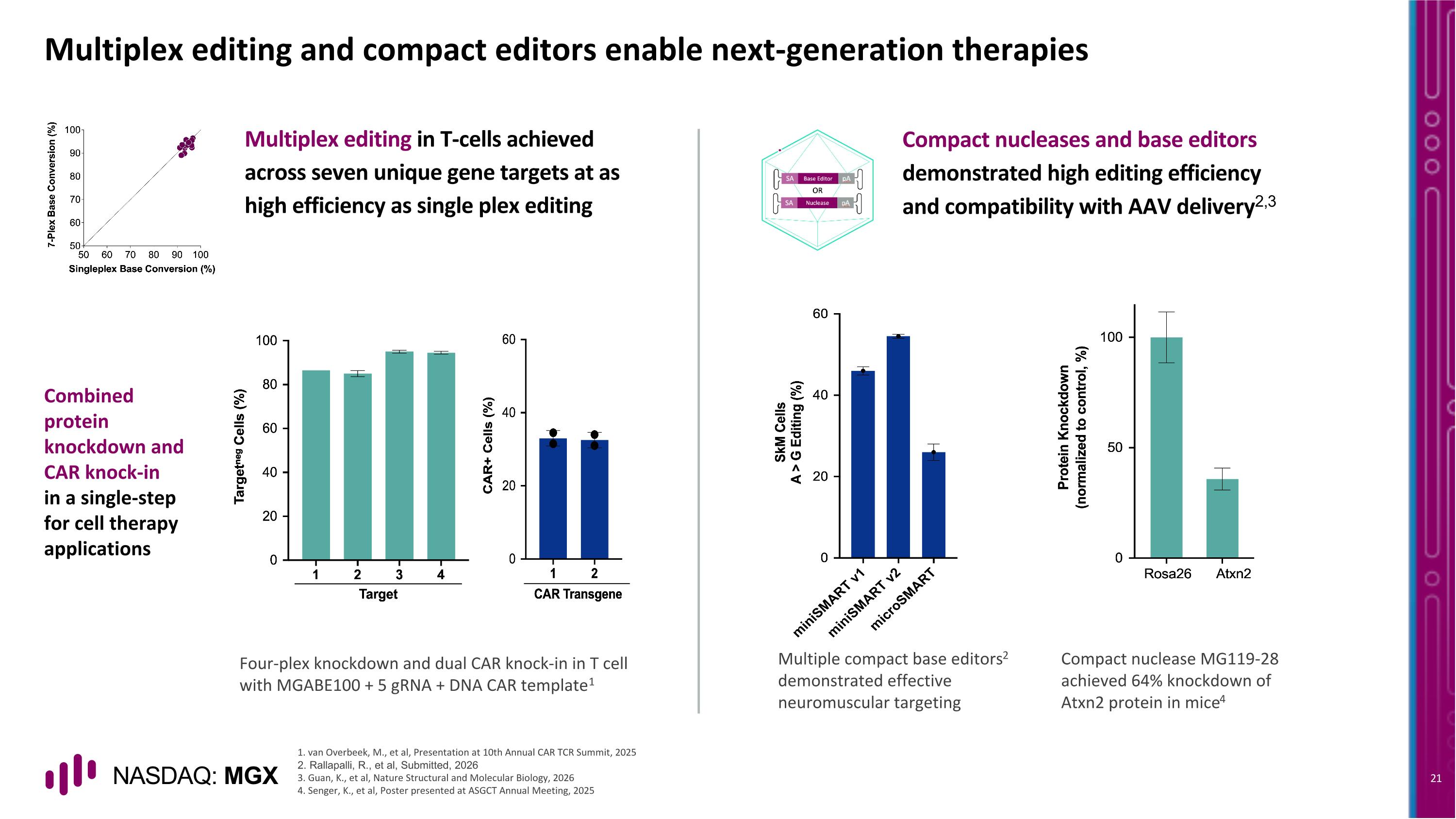
Multiplex editing and compact editors enable next-generation therapies Combined protein knockdown and CAR knock-in in a single-step for cell therapy applications Compact nucleases and base editors demonstrated high editing efficiency and compatibility with AAV delivery2,3 Four-plex knockdown and dual CAR knock-in in T cell with MGABE100 + 5 gRNA + DNA CAR template1 Multiple compact base editors2 demonstrated effective neuromuscular targeting Compact nuclease MG119-28 achieved 64% knockdown of Atxn2 protein in mice4 Multiplex editing in T-cells achieved across seven unique gene targets at as high efficiency as single plex editing 1. van Overbeek, M., et al, Presentation at 10th Annual CAR TCR Summit, 2025 2. Rallapalli, R., et al, Submitted, 2026 3. Guan, K., et al, Nature Structural and Molecular Biology, 2026 4. Senger, K., et al, Poster presented at ASGCT Annual Meeting, 2025

Strategic partnership expands reach into large cardiometabolic markets TTR AGT APOC3 Undisclosed Current indications: MGX's in vivo genome editing complements Ionis leadership in cardiometabolic space 4 targets: two co-development and co-commercialization options Multibillion dollar TAM

Building a leading gene editing company focused on cures Broad and differentiated library of proprietary gene editing technologies representing a significant long-term value driver Advancing MGX-001 for hemophilia A with clear development path and well-defined clinical and regulatory endpoints Extending beyond hemophilia A, large gene integration system opens potential to address other protein deficiencies Pairing our gene-editing capabilities with complementary expertise to accelerate development through collaboration 23

Thank you

























